Karlsruhe: First implantation of an extravascular defibrillator
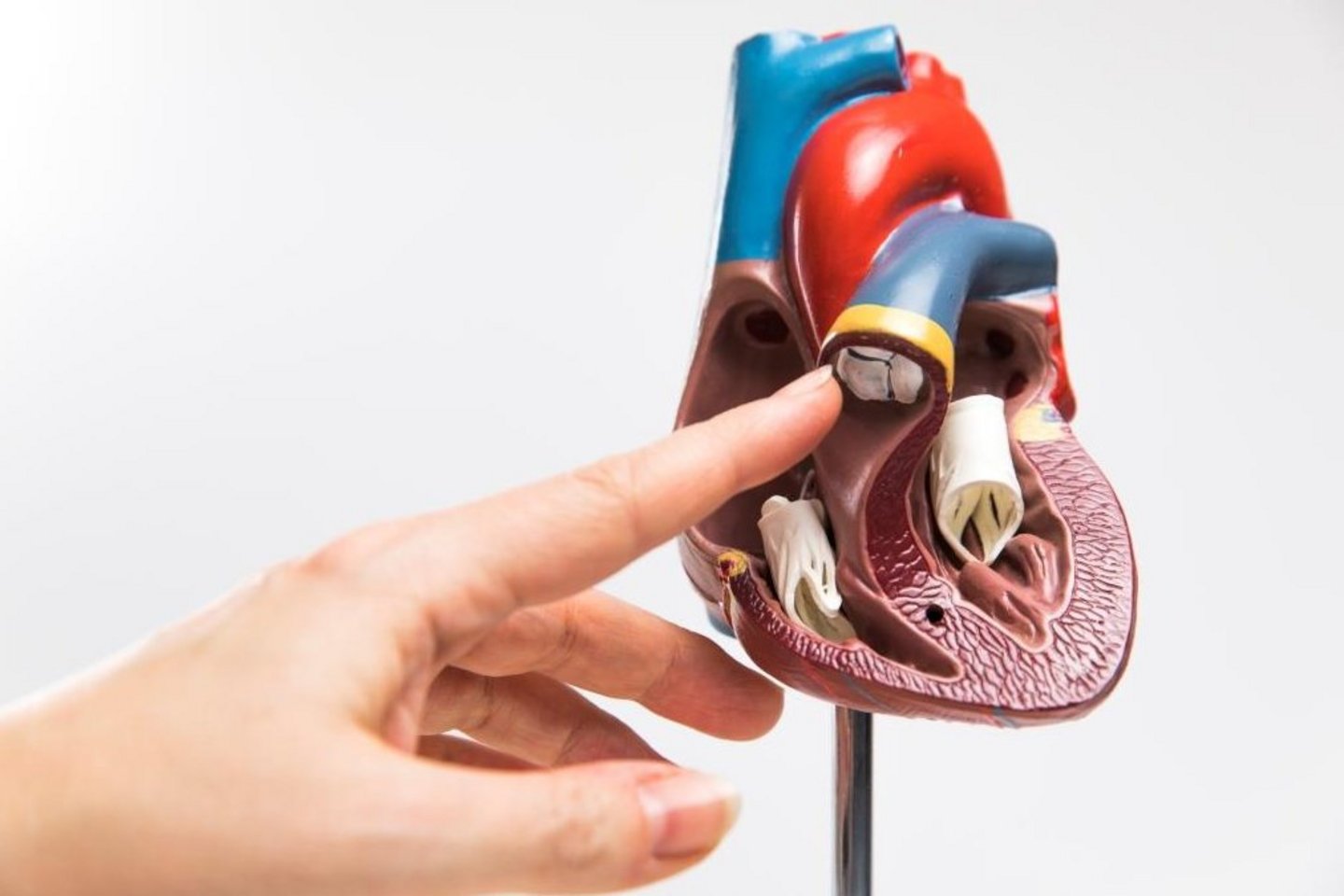
If patients suffer from malignant cardiac arrhythmias such as ventricular fibrillation or an excessively high heart rate (ventricular tachycardia), the heart can no longer pump sufficient blood throughout the body. As a result, organs are deprived of oxygen. Symptoms range from dizziness and shortness of breath to loss of consciousness. To prevent sudden cardiac death, heart rhythm must be continuously monitored and restored as quickly as possible using a defibrillator.
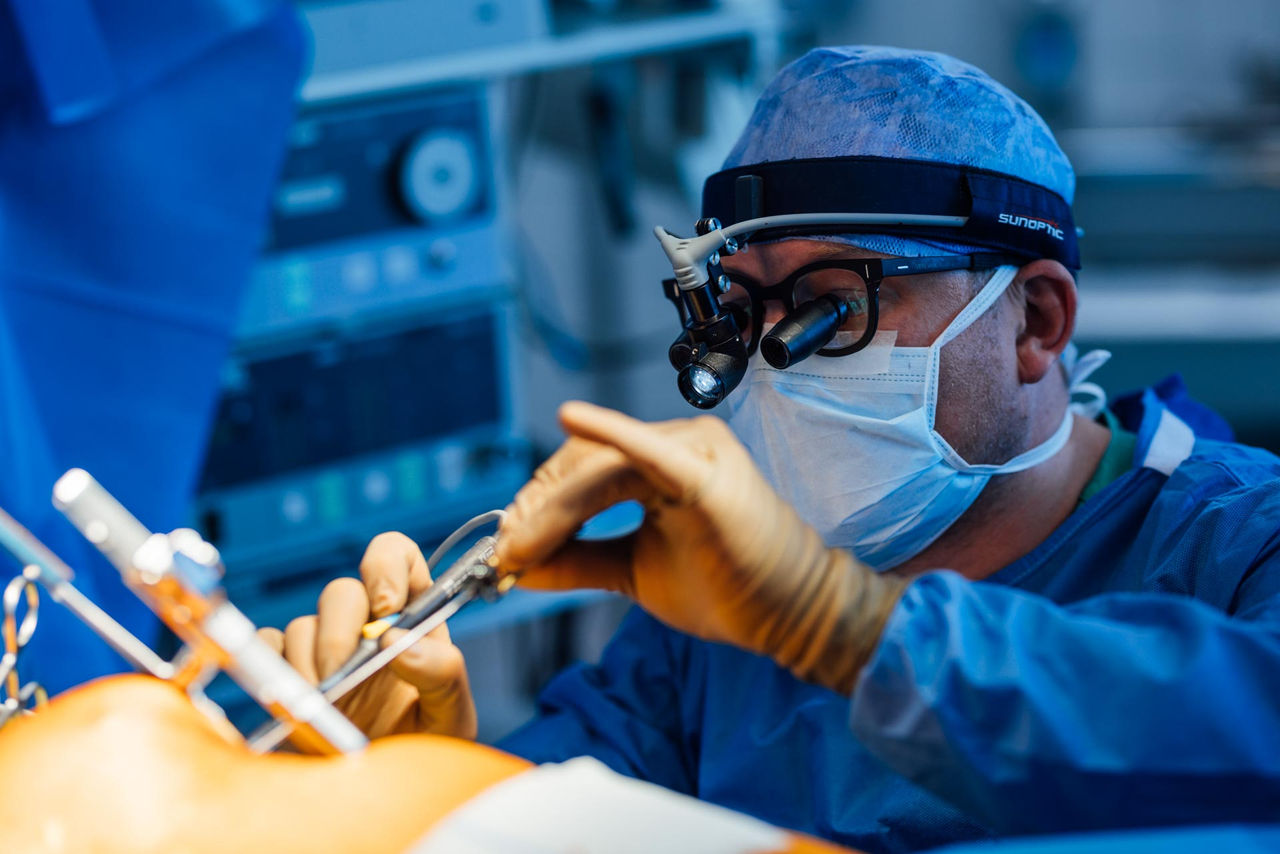
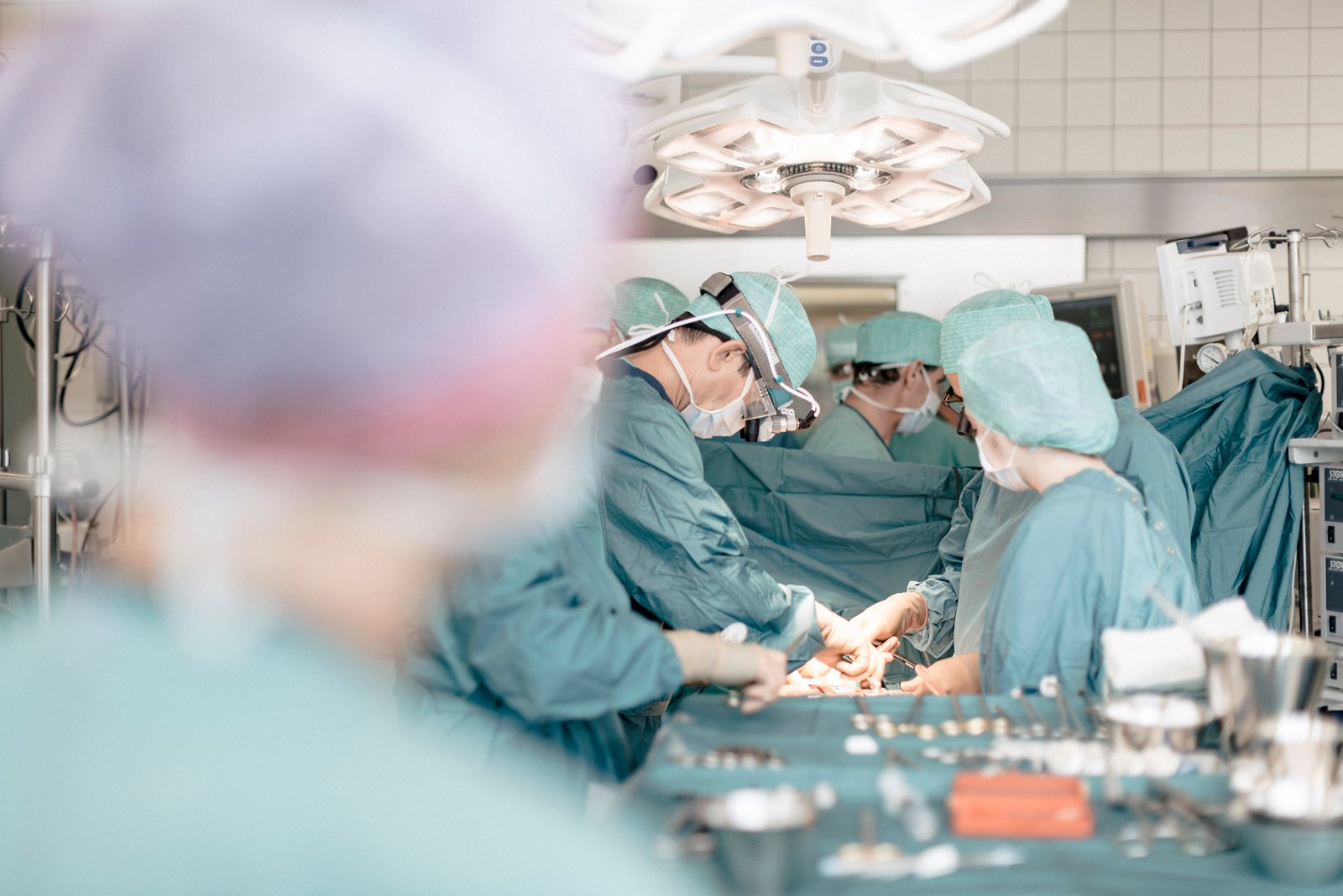
To treat such life-threatening arrhythmias, rhythm surgery specialists at the Heart Surgery Hospital Karlsruhe have for the first time implanted a so-called extravascular defibrillator (EV-ICD). This device combines the functions of a conventional transvenous defibrillator (TV-ICD) with the advantages of a subcutaneous defibrillator (S-ICD).
Like a transvenous defibrillator, the extravascular defibrillator has a pacing function. This means it can act as a pacemaker by delivering electrical impulses. It can compensate for prolonged pauses in the heartbeat and also provide antitachycardia pacing (ATP) to control excessively high heart rates. Only if arrhythmias persist despite gentle electrical impulses does the device deliver a shock.
This additional pacing capability represents an advancement over the subcutaneous defibrillator, which can only deliver shock therapy. Therefore, the S-ICD is mainly suitable for patients who do not require pacing and who have already undergone heart surgery. Both systems share a lower risk of complications. Unlike transvenous defibrillators, where at least one lead is inserted into the right ventricle via a vein, both subcutaneous and extravascular defibrillators are placed outside the blood vessels and the heart. This reduces long-term risks such as vascular occlusion, infections, and heart valve dysfunction. It also lowers the risk of lead fractures, infections (endocarditis), and complications during lead extraction.
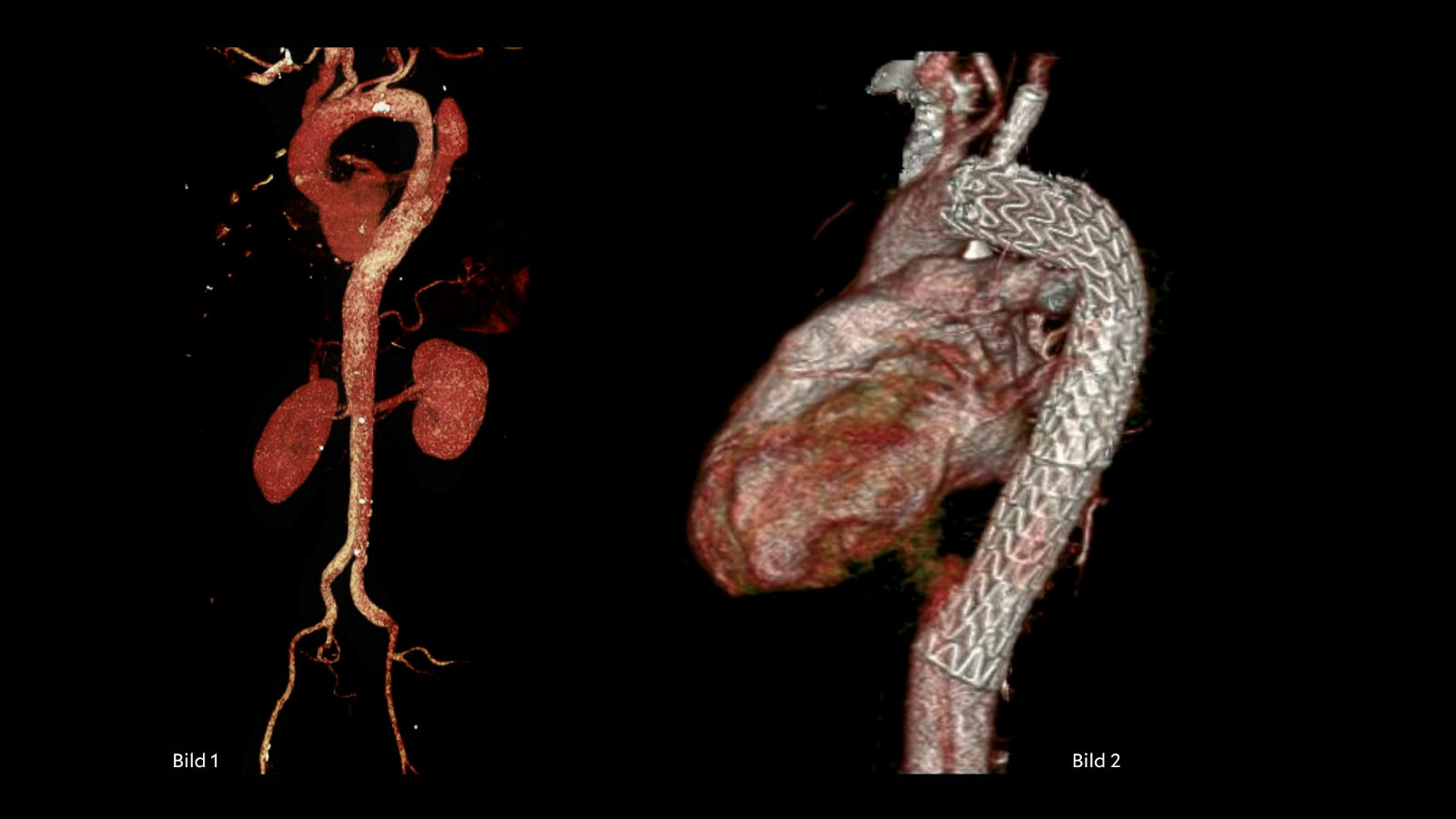
While in subcutaneous defibrillators the electrode is positioned above the sternum, in extravascular systems it is placed substernally (beneath the sternum) and lies on the pericardium, essentially directly on the heart.
“The close proximity to the heart represents a real revolution in device therapy. The extravascular defibrillator is the best option for patients without prior heart surgery, with normal sinus rhythm, and with appropriate heart rate response to physical activity. Patients benefit from the functions of conventional defibrillators while facing fewer complications, since no leads need to be inserted into the heart. In addition, arrhythmias can often be treated without painful shocks, significantly improving patient comfort,” explains Dr. Ghaffari Mehrjerdi.
Prof. Dr. Jürgen Kuschyk from the University Hospital Mannheim adds: “After more than 20 years of development, the extravascular defibrillator represents a new milestone in cardiac device therapy. Due to the difficult access to the space between the sternum and the heart, it was previously not possible to place the electrode beneath the sternum instead of inside the heart. Now we can avoid intracardiac leads altogether, which is a true game changer, especially for patients with high surgical risk, susceptibility to infections, or valve disease.”
The innovative device was evaluated in a global clinical trial involving 356 patients at 46 centers in 17 countries. The defibrillation success rate was 98.7%. The battery life is estimated to last up to 11.7 years. CE certification was obtained in 2023, and early implantations worldwide have already shown promising results. “I am very pleased that we successfully treated our first patient with this advanced device. I am confident it will help save many more lives,” says Prof. Dr. Uwe Mehlhorn.
Site Director Dominik Teich adds: “We are proud to be the first clinic in our region to offer this life-saving and minimally invasive procedure. Our expertise allows us to act as pioneers within the Helios group.” At the Helios Heart Center Leipzig, specialists are currently undergoing intensive training to also offer this procedure in the future. The program is expected to launch in October 2025.
Helios Heart Surgery Hospital Karlsruhe
The Helios Heart Surgery Hospital Karlsruhe has been part of the Helios network since 2014. Founded in 1995, it has 96 beds, including 24 intensive care and 16 intermediate care beds. With more than 1,800 procedures annually, it is one of the largest cardiac surgery centers in Germany.
Helios is part of the Fresenius Group and is Europe’s leading private healthcare provider, employing around 128,000 people. Approximately 26 million patients receive treatment annually. In 2024, the company generated over €12.7 billion in revenue.
Read more:
Do you need more information about Helios Hospitals or want to schedule your treatment?